The long-term financial health of a clinical research site depends on prudent management of its opportunities and resources. It is no secret that a large percentage of Principal Investigators discontinue conducting clinical trials each year for financial reasons.
A number of factors contribute to the financial woes clinical research sites experience. Some of the major problems sites face includes:
- accepting inadequate budgets
- incurring unanticipated expenses
- having an insufficient number of studies to generate adequate revenue
- inadequate cash-flow due to lengthy payment terms
- High risk financial dependencies
This post addresses the last item in the list above. In addition to understanding its costs and negotiating reasonable budgets and contract terms, sites should establish target financial metrics and then strive to operate under the principle of the “Rule of Tens™.”
Conceptually, the Rule of Tens is simple. In practice, it requires constant monitoring and diligence to achieve. When achieved for each metric, a site will have a solid financial foundation and the strength to withstand many of the financial vagaries of this industry.
The Rule of Tens, created by John Neal, CEO of PCRS Network™, has been used successfully by many sites. So what is the Rule of Tens?
At its core the Rule of Tens establishes maximum risk thresholds for critical metrics at no more than ten percent of the total being measured. It acts as an early warning system when a key metric begins to creep out of the target range.
In application, no more than ten percent of the total key metric (revenue, receivables, payables, etc.) should be related to a single source. A site increases its risk any time they become dependent on any one sponsor, CRO, broker, vendor, or have a concentration of studies within a single indication.
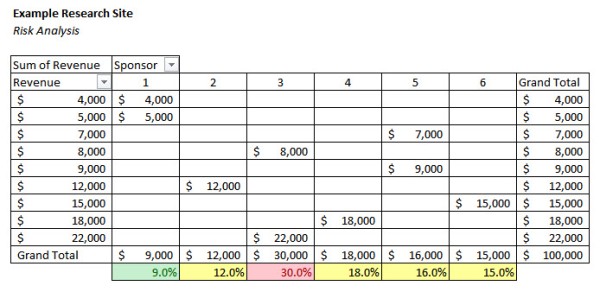
A simple example of a Rule of Tens analysis is below.
This analysis evaluates the relative risk from a concentration of study revenues with specific sponsors. Based on the Rule of Tens, revenue related to Sponsor 1 is below the ten percent threshold, so it represents an acceptable level of risk. Sponsor 3 represents thirty percent of the sites total revenue. This level of concentration places the site at significant risk. If this sponsor withdrew its studies, decided not to consider the site for future studies, ran into financial difficulty, or simply slowed down payments to the site, the site could experience serious financial difficulties.
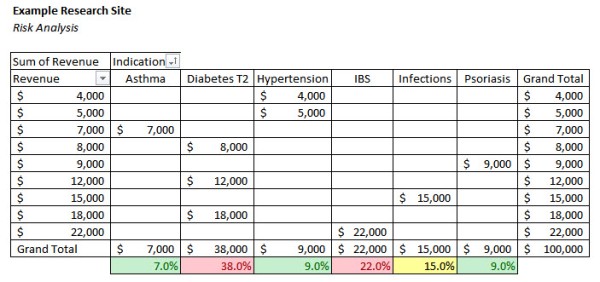
The example below uses the same data set but analyzes the risk of concentration by indication.
In this example, Asthma, Hypertension, and Psoriasis are below the ten percent risk threshold. The site is at serious risk with 38% of its revenue coming from Diabetes T2 studies and 22% from IBS studies. The site should take steps to diversify the areas in which it conducts studies and reduce its dependence on indications above the established risk threshold.
Reducing your concentration in a metric should not imply reducing the amount of studies accepted from a particular sponsor, or declining studies in a highly concentrated indication. Instead, it points to the need to expand other relationships or increase the range of indications the site conducts.
When a site first establishes the Rule of Tens analysis and discipline, the challenge to bring risky concentrations down to acceptable levels can seem daunting. Most sites find they are at risk with concentrations in multiple areas. Bringing all risks from concentrations within the acceptable risk threshold will not be achieved overnight; it requires focused, sustained effort over a period of time.
You may choose to establish a different threshold than ten percent. What is important is that you evaluate your risks, establish thresholds that bring key metrics within acceptable levels, and then consistently measure and take action to reduce risky concentrations. Sites that employ this approach consistently are much more likely not only to survive, but to thrive in the long-term.
Copyright © 2014, PCRS Network. ALL RIGHTS RESERVED WORLDWIDE